Making the Built Environment
Manufacture of paper, plastics, chemicals and metals produce 71% of toxic emissions in the U.S.1 Materials were produced by nature; timber, rock, clay, reeds for thatch, hide, wool, silk, and bone. Then man began to make things and it labeled the times; the Stone age, the Bronze age, the Iron age, fantastic plastic age, and now the age of silicon.


Nature has four laws for manufacture.
- Life friendly process
- Ordered hierarchy in structure.
- The elements self assemble
- Template crystalline forms with proteins.
For millennia we made things out of natural materials and once they out lived their useful life it mattered little if they were discarded to decay and absorb into another life. The modern synthetic materials have a hard act to follow. The iniquitous prizing open of an abalone shell reveals a mother of pearl lining on a substance twice as tough as any high tech ceramic. A spider’s web weight for weight is four times stronger than steel. The adhesion a mussel exudes to fasten itself to rock or reef in salt water needs no preparation and sticks to anything. Rhinoceros horn is capable of repairing itself yet contains no living cells. Add to that list bone, wood, skin, wool, tusk, antler, tendon and heart muscle. I have to ask; why compete?

Modern material extraction, processing and production of consumer goods have a commercial expense but add to that the rehabilitation of habitat from terra-forming and the toxicity of their waste accumulate an immense cost.

Ordered hierarchical structure
Biomimetics is either emulating or duplicating biosystems using mostly synthetic compounds and following traditional molecular chemical procedures2-4. With development in molecular5,8 and nanoscale6 engineering biomimetics has entered the molecular realm. Using natural molecular processes with nanoscale constructs molecular biomimetics is using hybrid methodology.7
Characteristic properties to be considered are; mechanical in nano-structured composites, magnetic properties of single domained particles, electro-conductivity of low dimensional semi-conductors, and properties in solution of colloidal suspensions10,11.
The control of nanostructures and ordered assemblies of materials in two and three dimensions still remains elusive 8 -11. Natural biomaterials are highly organized and often hierarchical in structure, with intricate nano-architecture that make a variety of functional soft and hard tissues. Rich Humbert and Mehmet Sarikaya of Uni. Of Washington have studied abalone nacre as a structural crystalline material.
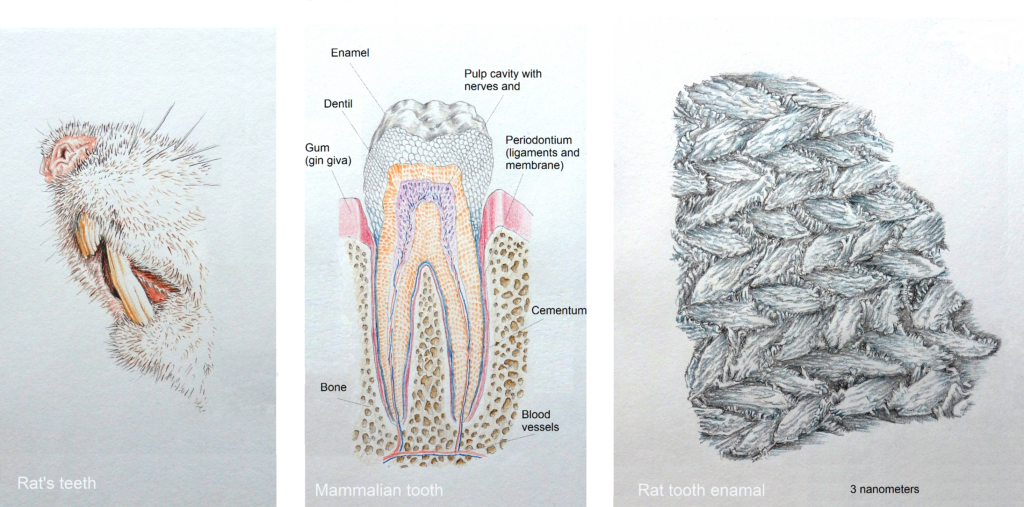
Illustrations follow of biologically synthesized complex materials with details as you would see with a scanning electron microscope.
Humbert and Sarikaya also compared tendons for their elasticity and rat’s tooth for toughness.
In aquatic environments tissues are synthesized where physical conditions are moderate. The animal producing the tissue provides genetic input through its DNA and more likely physiological direction. The natural choice of molecule evolved by selection as a protein. Their molecules have a large carbon based chain where other molecular groups attach in various arrangements to the chain. Other carbohydrates and lipids can do the structural building. Proteins are versatile they collect and transport raw materials and they assemble molecular units, for example amino acid groups. Into ordered nuclei and substrates3,4,12. This is probably achieved through the excitation of electron revolving around nuclei attaining higher energy state and changing position to align with other compatible nuclei.
Engineered materials are made using a combination of processes like melting, solidification, thermo-mechanical working. Fire and temperature treatments are both energy demanding procedures. Or place materials in solution and apply vacuum deposition and growth processes16,17.
In total contrast biological systems have lengthy evolutionary selection processes which has given appropriate molecular recognition to an ordered process to build as required by the organism18,19.

Using biomimetic processes, hybrid materials could potentially be assembled from molecules using recognition abilities of proteins. As it is we know proteins assemble molecular units that fit by way of attraction of opposite charged sites and repulsion of like charged sites along the protein chain. An inorganic surface-specific polypeptide could serve as a binding agent to control the organization and acquire the desired functions of material parts.
The building of combined functional structures is possible with biomimetics. Genetic DNA coding dictates the molecular pattern in the protein that then becomes the template for building. Proteins can be used as adhesives to link synthetic units like nano-particles, polymer or other molecular structures into molecular templates. Given suitable physical and chemical opportunity biological molecules assemble themselves into an ordered structure as seen in natural structures, then possibly a hierarchical structure as seen in the shells of mollusks.
Rich Humbert and Mehmet Sarikaya of Washington University have studied the nacre(mother –of-pearl) of the abalone shell an incredibly tough structure, tendons are extremely pliable and strong and the impact resilient rat’s tooth as examples of high caliber protein based materials.
Present understanding of repetitious folding of protein molecules and surface adhesion chemistry is sufficient to deliver design of proteins20. To work around the choice of complex proteins data bases of generated peptides can be perused for binding properties to inorganic surfaces by using phage (eating and combining) and cell-surface display techniques21. ( see Further Reading below ) In the future it is plausible to facilitate a molecular erector manufacturing kit to make proteins designed to link to a specified inorganic surface that could self assemble into intricate structures composed of inorganics and proteins22.
Scientist like Paul Calvert of Arivona University, Materials laboratory in Tuscan “ … want to grow lattices with perfection in crystal size, shape, orientation and location especially in the ceramic world of insulation, bearings, wear and temperature resistant coatings and where specific optical, electrical and chemical requirements are needed. In the nano-world brittleness is the main issue.”23
Bones are crystals of calcium phosphate deposited in a polymer matrix. Diatoms – micro sea creatures – look like snowflakes, have skeletons made of silica glass shaped by the organic membrane of their bodies. Teeth in general, the spines of sea urchins, the shells of sea snails are very hard24, and are all formed of inorganic crystalline structures. The lamprey’s teeth are extra hard crystals to gnaw through rock. A bacterium Magnetotactic has managed to grow crystals of iron-oxide – magnetite in micro vesicles(balloons) inside its body.25 The magnetite vesicles line up in a chain assisting the bacteria to orientate itself toward the magnetic center of the planet, the anaerobic zone where they find their food source in bottom sediment26.

The abalone emits a thin smear of polymer in its shell this matrix mortar is made of polysaccharides(sugars). Then the outer layer cells of the mollusk secret an organic mix in which the shell matrix proteins determine the mineralogical and crystallographic make up of the shell. This enables the formation of the hexagonal units within the shell structure27.

Counter intuitive ordering of Structure
The Haliotidae family abalone’s cells secrete proteins and polysaccharides (other species may use lipids) into the fluid medium surrounding them. These polymers self assemble into three dimensional compartments that define the space to be mineralized. In the abalone’s case compartment after compartment is formed, layer upon layer is applied each off-set from the lower to achieve overlapping and interlocking structure.
Inside each compartment seawater – saturated with calcium ions and carbonate ions – eventually making a calcium carbonate crystal. The ions being charged are attracted to oppositely charged chemical groups protruding from the walls of the compartments. When the first layer of ions settle the pattern is established for the whole crystal.
The mollusk releases template proteins into the compartments. The protein polymers self assembly to line the walls in an array of negatively charged landing sites awaiting the positively charged ions in the sea water.
Protein molecules are very long chains with hundreds of amino groups. Each amino acid has a differing charge arrangement and those various charge arrangements pull the prtein to fold in a preferred shape when floating in the cell’s fluid.
The folded arrangement of the protein influences the amino acid behavior in water. Neutral, water repelling amino acids burrow into the center of a protein complex. Charged, water attracting amino acids go to the periphery. The amino acid groups interact similarly with one another, some repelling, some attracting to bond, What eventually develops is a form uniquely suited to its function.
A protein may have a structural role like assembling tissue or bone others may have a function like hemoglobin, insulin, neuron receptors, antibodies, enzymes(orchestrate and speed up chemical reactions), having shapes ideally suited to their task.
For the abalone’s shell the protein chain folds into regular zigzag shape which bonds with another ‘saw-tooth’ protein to form a piano accordion like pleated sheet. One face of the sheet sticks out into the room, the other bonds with the floors, walls and ceiling surfaces of the compartments. Daniel Morse, director of Marine Biology Center at the University of California, Santa Barbara has determined that the groups that bond to the walls are neutral (glycine and alanine) and those projected into the space are negatively charged (aspartate). The bond points (landing sites) on the pleats are defined by the folded points of the protein molecules which is regulated by the position of the amino acids every few nanometers. The Sea solution makes the charged ions float onto the oppositely charged bonding points of the pleats.

The arrangement of the first layer of ions determines the pattern of the crystal growth – rhomohedral in the case of the nacre(inner shell surface) – prismatic crystals form the outer shell. The crystal shape and its orientation gives the material qualities of light reflection and absorption or conductivity or hardness. Fourteen different crystal shapes have been found in natural materials.
Self Assembly
If you could apply a selection of protein into a cavity, allow the protein molecules to fold and layer the walls of the space. You could pump a solution of the desired ions into the cavity and the crystals would self-assemble to form the desired coating of the fourteen known kinds. This assembly is enabled by like charges repelling and opposite charges attracting. The electrostatic attraction holds the molecules together until more permanent bonds are locked into the key arrangements by catalysts like enzymes. All this assembly can only take place if molecules are available usually in a free moving fluid like air or water.
Templates
Template is a term for the appropriate sequenced protein that will orchestrate positioning and alignment of constituent molecular parts. The prime candidate for making them is E. coli bacteria. Humans have used bacteria, yeast, moulds for making beer, wine, bread, and matured cheese. Today it is used industrially for food additives, antibiotics, chemicals and vitamins.
This topic continues in Designing from Nature’s Template {hyperlink here} the next Living Design Article.
Further Reading
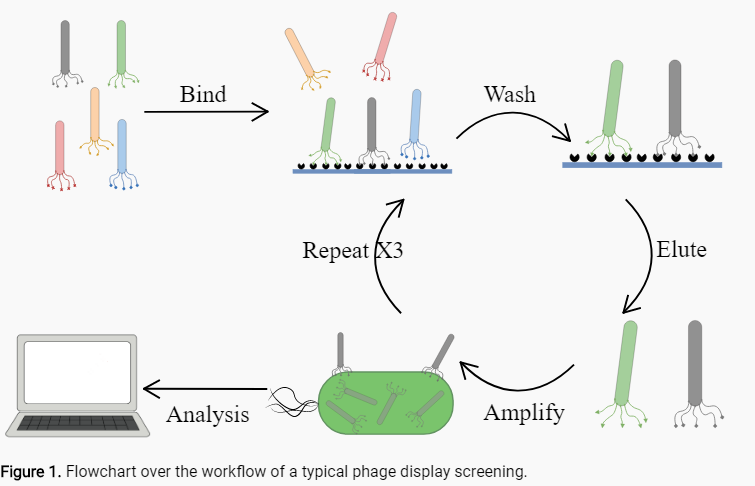
Phage display is a laboratory technique for the study of protein–protein, protein–peptide, and protein–DNA interactions that uses bacteriophages (viruses that infect bacteria) to connect proteins with the genetic information that encodes them. In order to determine a peptide with affinity to a molecule of interest an iterative process of affinity binding and washing, called panning, is needed to yield an end result with a high concentration of high affinity peptides28.
Referenced footnotes
Ref 1: Efficiency Revolution: Creating a sustainable materials economy. J.E. Young & A.Sachs.
Ref 2: 5. Mann, S. & Calvert, P. Synthesis and biological composites formed by in-situ precipitation. J.Mater. Sci. 23, 3801–3815 (1988).
Ref 3: 6. Sarikaya,M.& Aksay, I. A. (eds.) Biomimetics: Design & Processing of Materials (American Institute of Physics,New York, 1995).
Ref 4: 7. Mann, S. (ed.) Biomimetic Materials Chemistry (VCH,New York, 1996).
Ref 5: 9. Drexler, K. E.Nanosystems (Wiley Interscience,New York, 1992).
Ref 6: 13. Ball, P. Life’s lessons in design. Nature 409, 413–416 (2001).
Ref 7: 14. Ferry,D. K. & Goodnick, S. M. Transport in Nanostructures (Cambridge Univ.Press, Cambridge, UK, 1997).
Ref 8: 15.Harris, P. J. Carbon Nanotubes and Related Structures (Cambridge Univ. Press,Cambridge, UK, 1999).
Ref 9: 16. Bachtold, A.,Hadley, P.,Nakanishi, T. & Dekker,C. Logic circuits with carbon nanotubes transistors. Science 294, 1317–1320 (2001).
Ref 10: 17. Gittins,D. I., Bethell,D., Schiffrin,D. J. & Michols, R. J. A nanometre-scale electronic switch consisting of a metal cluster and redox-addressable groups.Nature 408, 67–69 (2000).
Ref 11: 18.Muller, B.Natural formation of nanostructures: from fundamentals in metal heteroepitaxy to applications in optics and biomaterials science.Surf. Rev. Lett.8, 169–228 (2001).
Ref 12: 20. Lowenstam, H. A. & Weiner, S. On Biomineralization (Oxford Univ. Press,Oxford, UK, 1989).
Ref 13: 21.Mayer, G. & Sarikaya,M.Rigid biological composite materials: Structural examples for biomimetic design. Exp.Mech. 42, 395–403 (2002).
Ref 14: 22. Fong, H., Sarikaya, M.,White, S. & Snead, M. L.Nanomechanical properties profiles across DEJ in human incisor teeth. J.Mater. Sci. Eng C 7, 119–128 (2000).
Ref 15: 23. Sarikaya,M. et al. Biomimetic model of a sponge-spicular optical fiber-mechanical properties and structure. J.Mater. Res. 16, 1420–1428 (2001).
Ref 16: 24. DeGarmo, E. P., Black, J. T. & Kohner, R. A.Materials and Processes in Manufacturing (McMillan,New York, 1988).
Ref 17: 25. Sarikaya, M., Steinberg, B. & Thomas, G. Optimization of Fe/Cr/C base structural steels for improved strength and toughness.Metall. Trans. A 13, 2227–2237 (1982).
Ref 18: 26. Ponting,C. P.& Russell,R. R. The natural history of protein domains. Ann. Rev.Biophys. Biomol. 31, 45–71 (2002).
Ref 19: 27. Izrailev, S., Stepaniants, S., Balsera, M., Oono,Y.& Schulten,K.Molecular dynamics study of unbinding of avidin-biotin complex. Biophys. J. 72, 1568–1581 (1997).
Ref 20: 28. Schonbrun, J.,Wedemeyer,W. J.& Baker,D. Protein structure prediction in 2002.Curr.Opin. Struct. Biol. 12, 348–354(2002).
Ref 21: 29. Brown, S.Metal recognition by repeating polypeptides.Nature Biotechnol. 15, 269–272 (1997).
Ref 22: 11. Sarikaya,M. Biomimetics:Materials fabrication through biology. Proc.Natl Acad. Sci. USA 96, 14183–14185 (1999).
Ref 23: Janine Benyus, Biomimicry: Innovation Inspired by Nature. P99. W.Morrow (2002)
Ref 24: Craig Welch Sea change: vital part of food web dissolving. Seattle Times photos S.Ringman
Ref 25: Richard B. Frankel and Dennis A. Bazylinski: How magnetotactic bacteria make magnetosomes queue up. digitalcommons.calpoly.edu
Ref 26: R. P. Blakemore, R. B. Frankelt & Ad. J. Kalmijnt: South-seeking magnetotactic bacteria in the Southern Hemisphere. digitalcommons.calpoly.ed
Ref 27: Xiaorui Song, Zhaoqun Liu, Lingling Wang and Linsheng Song, Proteins and Cellular Orchestration in Marine Molluscan Shell Biomineralization, pub: Frontiers in Marine Science. 2019
Ref 28: 2018.igem.org/Team:Uppsala/Phage_Display

Love the site– really individual pleasant and great deals to see!